3D Printlife Copper 3D PLACTIVE AN1 Antimicrobial PLA Filament PLAC
$107.00
3D Printlife Copper3D PLACTIVE AN1 Antimicrobial PLA Filament PLAC Available in 1.75mm and 2.85 mm diameter and 4 color choices: Blue, Green, Purple and Red.
Description
PLACTIVE AN1 – Antimicrobial PLA Filament
FDA-compliant and ISO-certified, PLACTIVE AN1™ is an innovative Nanocomposite developed with a high quality PLA and a patented, scientifically validated and highly effective Nano-Copper additive.
Copper has been shown to have protective properties.
Print Settings:
Use standard PLA settings
Nozzle Temp: 190°C – 220°C
A heated bed is not required but set it to 60°C if available.

PLACTIVE AN1(tm) is an innovative Nanocomposite developed with a high quality PLA and a patented, scientifically validated and highly effective Nano-Copper additive. This unique combination of technologies brings the following characteristics to Copper3D products:
- Antimicrobial action that has been scientifically validated eliminating hundreds of fungi, viruses, bacteria and a wide range of microorganisms.
- Antimicrobial properties confirmed by two microbiology laboratories in Chile and USA.
- Clinically tested in prosthesis for amputees with excellent results.
PLACTIVE AN1(tm) is ISO 10993-10: 2010 certified for biocompatibility of Skin Contact/Mucose Contact.
PLACTIVE(tm) Applications
PLACTIVE AN1 is ideal for the manufacturing of medical applications where it’s dangerous to have bacterial contamination, such as postoperative prostheses, wound dressing and surgical equipment.
Antimicrobial action that has been scientifically validated eliminating hundreds of fungi, viruses, bacteria and a wide range of microorganisms.
· 10% of all worldwide patients admitted in a hospital will acquire at least one healthcare-associated infection (HAI)1· Annually in the US, 2 million of patients gets infected in hospitals2 with 90.000 deaths. (2)
· The estimated cost for the US Healthcare System is up to 45 Billion USD (2,3)
· 50% of HAI’s at worldwide level are attributable to bacterial contamination of Medical Devices (4)
· Greater than 40% of Amputees present dermic complication due to High Bacterial Burden of Prosthetics and Orthotics (5)
Antimicrobial properties confirmed by two microbiology laboratories in Chile and USA.
PLACTIVE™ is a FDA Registered Material and EU compliant (No. 10/2011, No. 1935/2004 and No. 2023/2006).
The manufacturer also has certification ISO 9001/2015 and is REACH compliant.
Clinically tested in prosthesis for amputees with excellent results. Ideal for the manufacture of other medical applications where it’s dangerous to have bacterial contamination, such as postoperative prostheses, wound dressing and surgical equipment.
The Nano-Additive mantains all the mechanical properties of the base PLA material.
Non-toxic product and environmentally friendly (biodegradable).
See more information here: Copper3D
Copper3D (www.copper3d.com) is a chilean company born in an academic process in the Master of Innovation at the Adolfo Ibañez University, the most important business school in LatAm. In this academic program, the founding team of this company met:
Andrés Acuña, Electronic Civil Engineer with 14 years of experience in the mining sector and specializations in automation of mineral processing.

Daniel Martínez, Physical Therapist & MBA, with 13 years of experience in management, marketing, innovation and new business models in the Healthcare industry.
Claudio Soto MD, Physician with vast experience in rehabilitation, regulatory aspects of Healthcare and process management in large health institutions.
We realized that more than 40% of amputees suffer some type of dermic disorders due to the use of their prostheses. This phenomenon is also observed in non-amputee patients who use orthoses. The reason? The high bacterial burden present in these medical devices, which in contact with the skin can cause dermatitis, folliculitis, fungal or bacterial infections, among others.
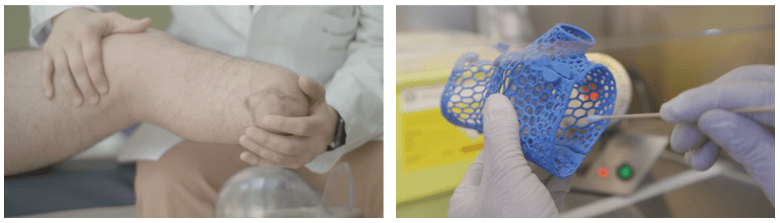
But then we realized that the problem of bacterial burden is much more complex and huge: According to some studies, about 50% of all HAI’s (Hospital Acquired Infections) worldwide are due to the bacterial burden of medical devices that are difficult to maintain clean and sterilyzed.
After a long process of research, development and innovation, we realized that the ideal solution to this problem involved the molecular intervention of the material with which these medical devices are manufactured. After discarding many options, we began prototyping a new polymer for 3D printing with an internationally patented additive containing copper nanoparticles, extremely effective in eliminating fungi, viruses and bacteria but safe for humans at the right concentrations.
With successful preliminary trials in Chile, we decided to start with the industrial manufacture of this material in Europe for later commercialization.
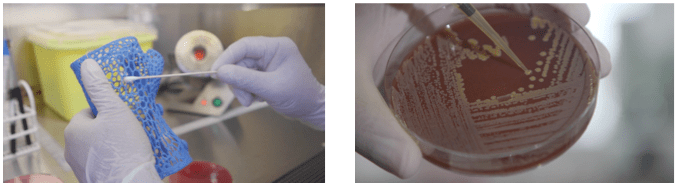
At the moment we have our first product available, it is a high quality PLA polymer with additive concentrations of 1 and 2%, it is called PLACTIVE since it is a PLA polymer with “active” properties in the elimination of a wide range of microorganisms.
We also developed a medical grade material called NANOCLEAN, it is a high quality PETG polymer with additive concentrations of 2 and 3% and aimed at more specific purposes in the world of medical devices.
We strongly believe in the impact that hard work, a great team and new technologies have on the quality of life of people, and in the power of innovation to reshape the future.
ANTIBACTERIAL 3D PRINTING, CLINICAL USE Nº10:
The Otoscope is one of the most used Medical Devices for its simplicity and versatility when it comes to examining ears, nose and throat.
But it has a weak point. The cone of the otoscope is the piece that comes into direct contact with the ear. It is a plastic piece (hence porous) that accumulates a high bacterial load if it is not cleaned or disposed of properly. This often causes patients (usually children) examined with contaminated otoscopes to leave the clinic / hospital with an otitis that falls within the group of Infections Associated with Health Care (IAAS).
You can find detailed instructions for its construction here, even with its lighting system:
https://www.wevolver.com/…/fie…/blob/master/documentation.md



CLINICAL USE Nº11:

ANTIMICROBIAL 3D PRINTED SURGICAL SEPARATOR Surgical separators are key pieces in any operating room in the world. Historically, they have been manufactured in stainless steel, but since it is a “passive” material, it allows to colonize quickly in the presence of even a minimal amount of microorganisms. A study published in the Journal of Surgical Research by the department of surgery of the U. of Arizona: (https://lnkd.in/dd_VNwA) concluded that these pieces can be perfectly manufactured with 3D printing, they take about 90 minutes to built, the final piece easily supports the forces necessary to fulfill its function within an operating room (+ 13Kg of Tangential Force) and only at ONE TENTH of the cost of the original piece of steel. If in addition to all these attributes, we add ANTIBACTERIAL and REUSABLE if manufactured with PLACTIVE OR NANOCLEAN (www.copper3d.com), so we have a new class of surgical, functional, cost-effective, in-situ manufacturing, customizable, reusable separators , that do not need sterilization and completely antibacterial since they are manufactured with an ACTIVE material. #Antibacterial3DPrinting
CLINICAL USE Nº 12
ANTIMICROBIAL 3D PRINTED SURGICAL SEPARATOR Surgical separators are key pieces in any operating room in the world. Historically, they have been manufactured in stainless steel, but since it is a “passive” material, it allows to colonize quickly in the presence of even a minimal amount of microorganisms. A study published in the Journal of Surgical Research by the department of surgery of the U. of Arizona: (https://lnkd.in/dd_VNwA) concluded that these pieces can be perfectly manufactured with 3D printing, they take about 90 minutes to built, the final piece easily supports the forces necessary to fulfill its function within an operating room (+ 13Kg of Tangential Force) and only at ONE TENTH of the cost of the original piece of steel. If in addition to all these attributes, we add ANTIBACTERIAL and REUSABLE if manufactured with PLACTIVE OR NANOCLEAN (www.copper3d.com), so we have a new class of surgical, functional, cost-effective, in-situ manufacturing, customizable, reusable separators , that do not need sterilization and completely antibacterial since they are manufactured with an ACTIVE material. #Antibacterial3DPrinting


1 World Health Organization (WHO)
2 Patricia W. Stone, Economic Burden of Healthcare-Associated Infections, An American Perspective.
5 (Dudek NL, 2005; Dudek NL, 2006; Maulenbelt HE, 2009)
Print Settings:
Use standard PLA settings Nozzle Temp: 190°C – 220°C,
A heated bed is not required but set it to 60°C if available.
Additional information
| Colors: | Blue, Red, Green, Purple |
|---|---|
| Diameter: | 1.75mm, 2.85mm |

















